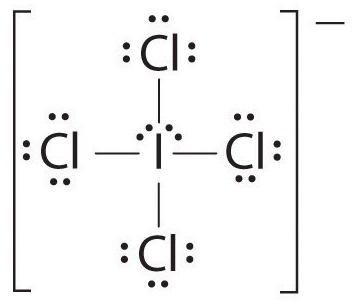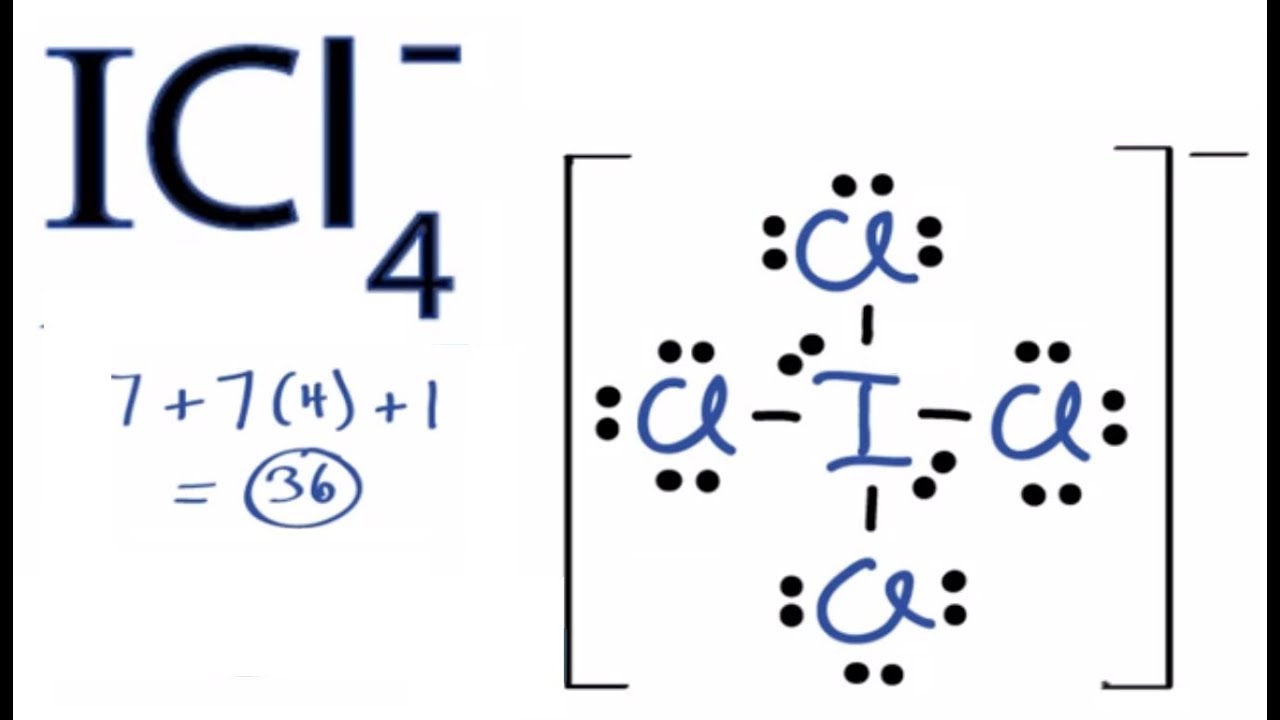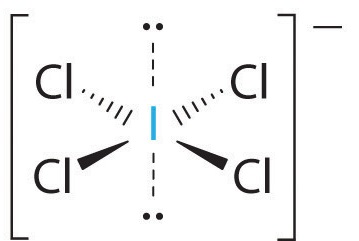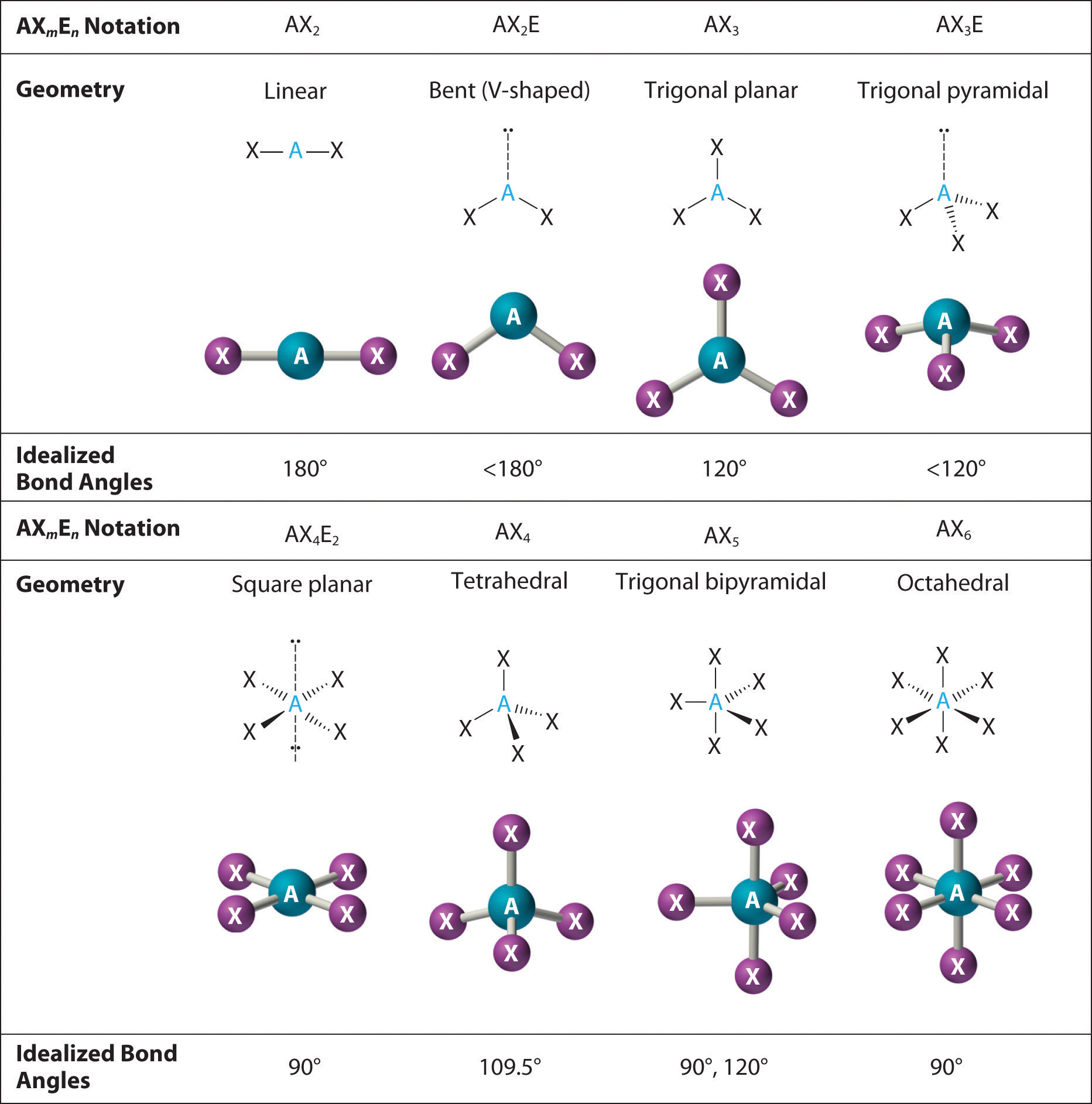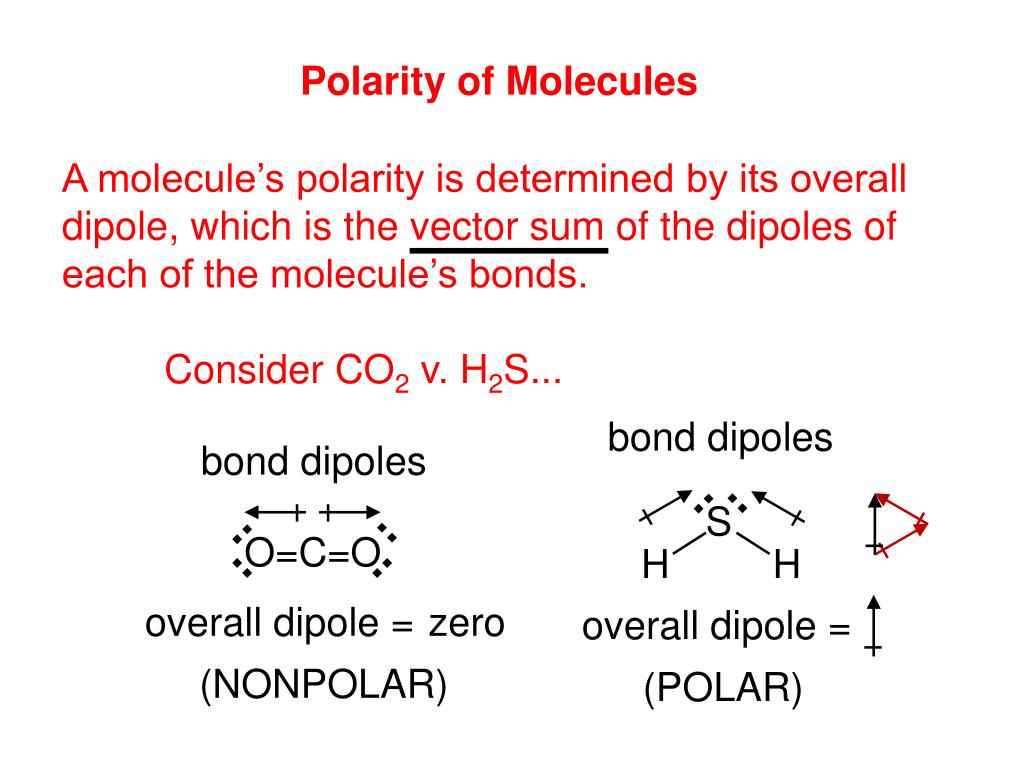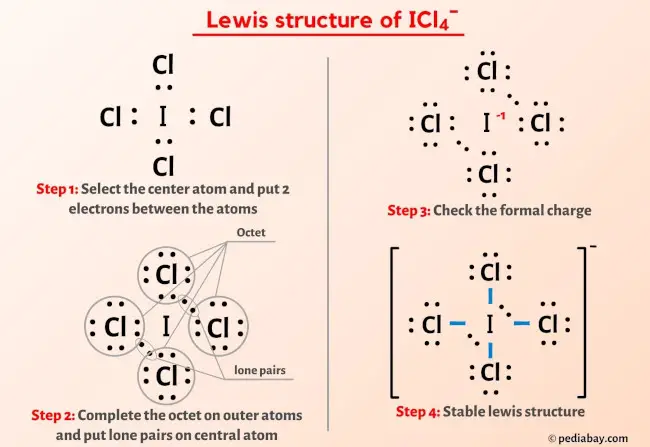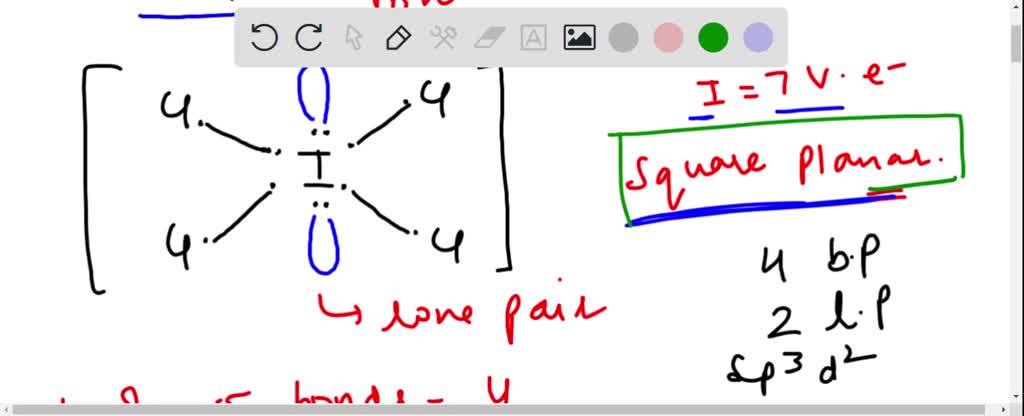
SOLVED: ICl4- Polar or Nonpolar VSEPR name: a. octahedral b. tetrahedral c. trigonal bipyramidal d. square planar Hybridization: a. sp2 b. sp3d2 c. sp3d d. sp3

SOLVED: a. What are the angles between nonbonding electron pairs in the following molecules? If there are none, write N/A. If there are multiple pairs, give all angles. 1. ICl4- 2. SH2

AX2E2 – Molecular geometry, Hybridization, Bond angle, Polarity | Molecular geometry, Molecular shapes, Molecular